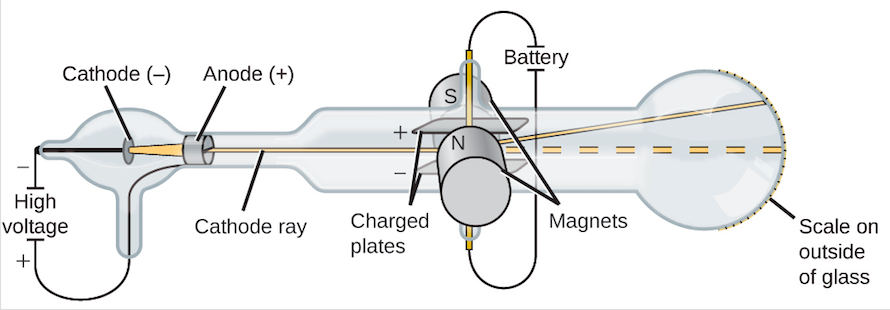
The English researcher, Ernest Rutherford, provided clearer focus when he bombarded a thin sheet of gold foil with alpha rays (beams of helium nuclei). Millikan, an American, devised an experiment showing that all electrical charges are integral multiples of a definite, elementary unit, which he quantified: electron charge:-1.6 X 10-19 C Still, scientists did not have a clear idea of what the atom looked like. Thompson, an English researcher, developed an experiment utilizing cathode rays to determine the charge to mass ratio of the electron. This led researchers to theorize that electricity consisted of units of charge that were particles (electrons), and that atoms contained such particles along with other oppositely charged particles (protons). It was discovered later that some compounds could be decomposed by electric current and that a certain quantity of electric current was required to decompose a certain amount of a particular substance (compound).
Dalton hypothesized that each certain element consists of small, identicle particles, which he called atoms, and explained the law of constant proportions by theorizing that atoms of particular elements combine in definite proportions to form specific compounds. The Development of the Atomic Theory of Matter Near the beginning of the 19th century, the law of simple multuple proportions was enunciated by the English physicist and chemist, Dalton, stating that the weights of the constituents (elements) of substances are in the ratio of small integers. The science of chemistry attempts to describe the properties of substances and the reactions that convert them into other substances. Matter assumes different forms called substances.

Our universe is formed of radiant energy and matter.
